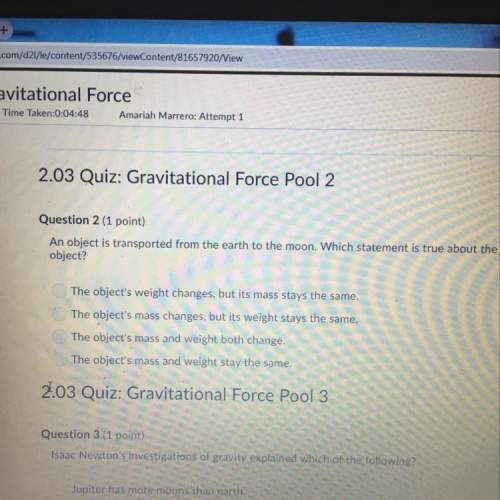
1) If there are 8.800x10^25 atoms of tin, how many moles is
this, and what would be the mass?<...

Chemistry, 12.03.2020 10:51 SoccerHalo
1) If there are 8.800x10^25 atoms of tin, how many moles is
this, and what would be the mass?
ii) You have a sample containing 2.575x10^24 molecules of
water. How many moles is this, and what is its mass?
Please help me I have no idea how to do this

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
The p sub shell can hold up to 8 electrons in an atom. true or false?
Answers: 1


Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

You know the right answer?
Questions


Mathematics, 01.10.2019 23:00

Social Studies, 01.10.2019 23:00


Mathematics, 01.10.2019 23:00









Chemistry, 01.10.2019 23:00

English, 01.10.2019 23:00



Biology, 01.10.2019 23:00

Mathematics, 01.10.2019 23:00

Spanish, 01.10.2019 23:00