
Chemistry, 12.03.2020 02:35 meganwintergirl
The partial pressures of CH4, N2, and O2 in a sample of gas were found to be 143 mmHg, 469 mmHg, and 563 mmHg, respectively. Calculate the mole fraction of oxygen. 20.1 0.399 0.741 0.479 0.359

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3



You know the right answer?
The partial pressures of CH4, N2, and O2 in a sample of gas were found to be 143 mmHg, 469 mmHg, and...
Questions


Mathematics, 05.05.2020 21:34

Mathematics, 05.05.2020 21:34

History, 05.05.2020 21:34

Mathematics, 05.05.2020 21:34

Mathematics, 05.05.2020 21:34

Mathematics, 05.05.2020 21:34



Advanced Placement (AP), 05.05.2020 21:34


History, 05.05.2020 21:34




Social Studies, 05.05.2020 21:34



History, 05.05.2020 21:34

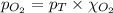
 = partial pressure of oxygen gas = 563 mmHg
= partial pressure of oxygen gas = 563 mmHg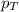 = total pressure = 1175 mmHg
= total pressure = 1175 mmHg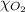 = mole fraction of oxygen gas = ?
= mole fraction of oxygen gas = ?


