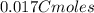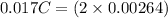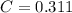Chemistry, 11.03.2020 22:15 hiiliohi1018
An aqueous solution of perchloric acid is standardized by titration with a 0.191 M solution of barium hydroxide. If 13.8 mL of base are required to neutralize 17.0 mL of the acid, what is the molarity of the perchloric acid solution?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:10
Which is true of transition metals when moving from left to right on the periodic table? the d sublevels are not filled across the period. the cation radii become larger across the period. atomic radii increase slightly and then start to decrease. atomic radii decrease slightly and then start to increase. o
Answers: 2

Chemistry, 21.06.2019 20:30
10. translate each of the following chemical equations into a sentence. a. 2 zns(s) + 3 o2(g) -> 2 zno(s) + 2 so2(g) b. cah2(s) + 2 h2o(l) -> ca(oh)2 (aq) + 2 h2(g)
Answers: 2

Chemistry, 22.06.2019 03:30
The boiling point of liquids is very high what does it indicate
Answers: 1

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1
You know the right answer?
An aqueous solution of perchloric acid is standardized by titration with a 0.191 M solution of bariu...
Questions



Mathematics, 01.06.2021 17:50



Mathematics, 01.06.2021 17:50

Mathematics, 01.06.2021 17:50

Mathematics, 01.06.2021 17:50

Mathematics, 01.06.2021 17:50




Mathematics, 01.06.2021 17:50

Social Studies, 01.06.2021 17:50

Mathematics, 01.06.2021 17:50

Biology, 01.06.2021 17:50


Mathematics, 01.06.2021 17:50

Mathematics, 01.06.2021 17:50

Mathematics, 01.06.2021 17:50

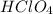 solution is 0.311 M
solution is 0.311 M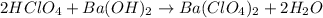
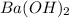 neutralizes 2 moles of
neutralizes 2 moles of 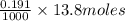 =
= 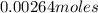
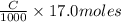 =
=