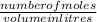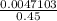A chemist prepares a solution of silver(I) nitrate AgNO3 by measuring out ×4.7102μmol of silver(I) nitrate into a 450.mL volumetric flask and filling the flask to the mark with water. Calculate the concentration in /mmolL of the chemist's silver(I) nitrate solution. Be sure your answer has the correc

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3

Chemistry, 22.06.2019 22:10
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1

Chemistry, 23.06.2019 06:10
2. what two items do autotrophs take from the environment to produce their food? 3. what are the two items that are released during transpiration from leaves? 4. what are the two membranes of the system? a.what are the two stages of photosynthesis? what are the two parts of photosynthesis?
Answers: 2

Chemistry, 23.06.2019 15:50
Many radioactive atoms that have large masses undergo radioactive decay by releasing a particle that is identical to a helium-4 nucleus. what changes in the original atom are expected as a result of this natural phenomenon? the atomic number and the mass number will decrease. the atomic number and the mass number will increase. the atomic number will increase, and the mass number will decrease. the atomic number will decrease, and the mass number will increase.
Answers: 2
You know the right answer?
A chemist prepares a solution of silver(I) nitrate AgNO3 by measuring out ×4.7102μmol of silver(I) n...
Questions


Chemistry, 14.02.2021 20:00

Computers and Technology, 14.02.2021 20:00


Social Studies, 14.02.2021 20:00


Mathematics, 14.02.2021 20:00

Mathematics, 14.02.2021 20:00

Mathematics, 14.02.2021 20:00



Mathematics, 14.02.2021 20:00



Mathematics, 14.02.2021 20:00


Mathematics, 14.02.2021 20:00

English, 14.02.2021 20:00


Mathematics, 14.02.2021 20:00