
Chemistry, 11.03.2020 13:33 lildanielmabien
Find the mass of AlCl3 that is produced when 25.0 grams of Al2O3 react with excess HCl according to the following equation.
Al2O3(s) + 6HCl(aq) → 2AlCl3(aq) + 3H2O(l)
Group of answer choices
72.9 g
155 g
65.4 g
16.3 g
32.6 g

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2

Chemistry, 22.06.2019 14:40
Pastoral farming is best described as a. a method of raising livestock and moving herds b. an african method of agriculture c. a method of cultivating crops on poor soils d. a common method of desert farming select the best answer from the choices provided a b c d
Answers: 2

Chemistry, 22.06.2019 17:30
Aroller coaster is traveling at 13 mi./s when you purchase a hill that is 400 m long and down the hill exonerate at 4.0 m/s squared what is the final velocity of the posterior found your answer to the nearest number
Answers: 1
You know the right answer?
Find the mass of AlCl3 that is produced when 25.0 grams of Al2O3 react with excess HCl according to...
Questions

History, 15.10.2019 23:30


Mathematics, 15.10.2019 23:30

Business, 15.10.2019 23:30

Biology, 15.10.2019 23:30

History, 15.10.2019 23:30

Mathematics, 15.10.2019 23:30

Mathematics, 15.10.2019 23:30






Mathematics, 15.10.2019 23:30






English, 15.10.2019 23:30

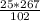 = 65.4 g of AlCl₃.
= 65.4 g of AlCl₃.

