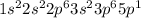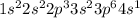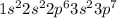Chemistry, 24.08.2019 03:50 SketchWasTaken
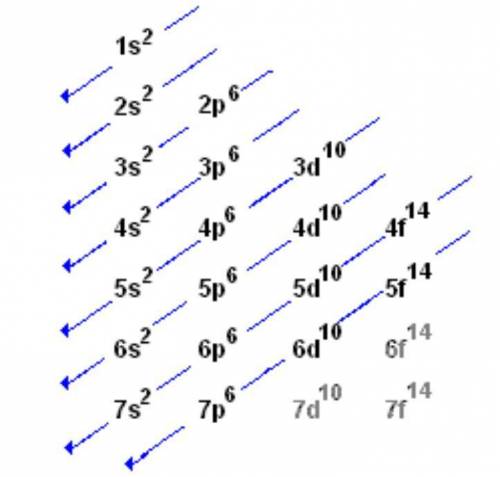
Which of the following is the ground state of an atom? which is its excited state? which is an impossible electron configuration? identify the element and briefly explain your choices. a. 1s^2 2s^2 2p^6 3s^2 3p^6 5p^1 b. 1s^2 2s^2 2p^3 3s^2 3p^6 4s^-1 c. 1s^2 2s^2 2p^6 3s^2 3p^7

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Problem #3 (ch. 1, problem 15)the ideal gas law provides one way to estimate the pressure exerted by a gas on a container. the law isí‘ťí‘ť=푛푛푛푛푛푛푉푉more accurate estimates can be made with the van der waals equationí‘ťí‘ť=푛푛푛푛푛푛푉푉â’푛푛푟푟â’푞푞푛푛2푉푉2where the term nb is a correction for the volume of the molecules and the term an2/v2is a correction for molecular attractions. the values of a and b depend on the type of gas. the gas constant is r, the absolutetemperature is t, the gas volume is v, and the number of moles of gas molecules is indicated by n. if n = 1 mol of an ideal gas were confined to a volume of v = 22.41 l at a temperature of 0â°c (273.2k), it would exert a pressure of 1 atm. in these units, r = 0.0826.for chlorine gas (cl2), a = 6.49 and b = 0.0562. compare the pressure estimates given by the ideal gas law and the van der waals equation for 1 mol of cl2 in 22.41 l at 273.2 k. what is the main cause of the difference in the two pressure estimates, the molecular volume or the molecular attractions?
Answers: 1

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 16:00
No copying 15 pts how does a free-body diagram tell you about the net force on an object?
Answers: 2

Chemistry, 23.06.2019 06:00
What volume of 0.500 mol/l hydrochloric acid, hci (aq) is required to react completely with 1.00 g of aluminum hydroxide, ai(oh)3 (s)?
Answers: 1
You know the right answer?
Which of the following is the ground state of an atom? which is its excited state? which is an imp...
Questions






Mathematics, 01.05.2021 16:50

Social Studies, 01.05.2021 16:50




Mathematics, 01.05.2021 16:50


History, 01.05.2021 16:50

Mathematics, 01.05.2021 16:50


Mathematics, 01.05.2021 16:50

Biology, 01.05.2021 16:50

Social Studies, 01.05.2021 17:00

Biology, 01.05.2021 17:00