In acidic solution, the breakdown of sucrose into glucose and fructose has this rate law: rate = k[H+][sucrose].
The initial rate of sucrose breakdown is measured in a solution that is 0.01 M H+, 1.0 M sucrose, 0.1 M fructose, and 0.1 M glucose.
How does the rate change if:
(a) [Sucrose] is changed to 2.5 M?
(b) [Sucrose], [fructose], and [glucose] are all changed to 0.5 M?
(c) [H+] is changed to 0.0001 M?
(d) [Sucrose] and [H+] are both changed to 0.1 M ?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:50
Problem page gaseous ethane reacts with gaseous oxygen gas to produce gaseous carbon dioxide and gaseous water . if of water is produced from the reaction of of ethane and of oxygen gas, calculate the percent yield of water. be sure your answer has the correct number of significant digits in it.
Answers: 2

Chemistry, 22.06.2019 06:30
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1


You know the right answer?
In acidic solution, the breakdown of sucrose into glucose and fructose has this rate law: rate = k[H...
Questions



Mathematics, 09.09.2021 03:30



Mathematics, 09.09.2021 03:30


Mathematics, 09.09.2021 03:30

Mathematics, 09.09.2021 03:30


Mathematics, 09.09.2021 03:30



Law, 09.09.2021 03:30

History, 09.09.2021 03:30


Biology, 09.09.2021 03:30


Mathematics, 09.09.2021 03:30

History, 09.09.2021 03:30

![[H^+]](/tpl/images/0542/2176/07acb.png) is changed to 0.0001 M than rate will be increased by the factor of 0.01.
is changed to 0.0001 M than rate will be increased by the factor of 0.01.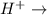 fructose+ glucose
fructose+ glucose![R=k[H^+][sucrose]](/tpl/images/0542/2176/8ca17.png)
![[H^+]=0.01M](/tpl/images/0542/2176/8ae83.png)
![R=k[0.01M][1.0 M]](/tpl/images/0542/2176/7a749.png) ..[1]
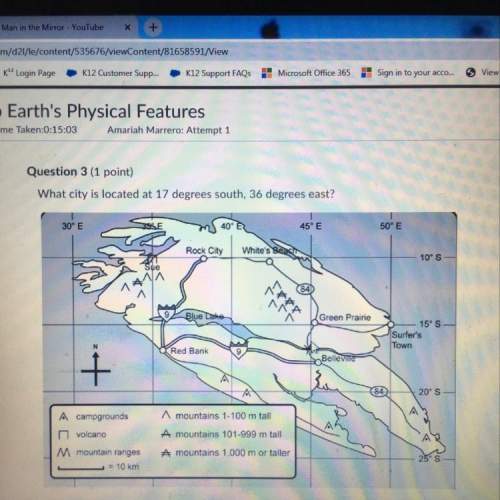
..[1]![R'=[0.01 M][2.5 M]](/tpl/images/0542/2176/5a418.png) ..[2]
..[2]![\frac{R'}{R}=\frac{[0.01 M][2.5 M]}{k[0.01M][1.0 M]}](/tpl/images/0542/2176/ac5ef.png)
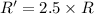
![R'=[0.01 M][0.5 M]](/tpl/images/0542/2176/d8698.png) ..[2]
..[2]![\frac{R'}{R}=\frac{[0.01 M][0.5 M]}{k[0.01M][1.0 M]}](/tpl/images/0542/2176/036aa.png)
![R'=[0.0001 M][1.0 M]](/tpl/images/0542/2176/62405.png) ..[2]
..[2]![\frac{R'}{R}=\frac{[0.0001 M][1.0M]}{k[0.01M][1.0 M]}](/tpl/images/0542/2176/7dc89.png)
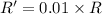
![R'=[0.1M][0.1M]](/tpl/images/0542/2176/67bb4.png) ..[2]
..[2]![\frac{R'}{R}=\frac{[0.1M][0.1M]}{k[0.01M][1.0 M]}](/tpl/images/0542/2176/6860b.png)