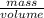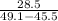Chemistry, 10.03.2020 23:07 hgdthbgjnb5604
A 28.5 gram piece of iron is added to a graduated cylinder containing 45.5 mL of water. The water in the cylinder rises to the 49.1 mark. Calculate the density of the iron piece.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 23.06.2019 01:00
How does carbon monoxide pose the greatest threat to humans? a. it can be produced by wood fires. b. it can be produced by home furnaces. c. it is produced by acid rain. d. it is produced by modern automobiles.
Answers: 2

Chemistry, 23.06.2019 03:00
Use the half-reactions of the reaction au(oh)3 + hi -> au +i2 +h2o to answer the questions
Answers: 1

Chemistry, 23.06.2019 08:20
At which temperature would a reaction with ah= -220 kj/mol and as=-0.05 kj/(mol-k) be spontaneous?
Answers: 2
You know the right answer?
A 28.5 gram piece of iron is added to a graduated cylinder containing 45.5 mL of water. The water in...
Questions



English, 31.01.2020 21:43

Mathematics, 31.01.2020 21:43

English, 31.01.2020 21:43

Mathematics, 31.01.2020 21:43

Health, 31.01.2020 21:43



Social Studies, 31.01.2020 21:43

Mathematics, 31.01.2020 21:43



Social Studies, 31.01.2020 21:43

Geography, 31.01.2020 21:43



Mathematics, 31.01.2020 21:43