Chemistry, 10.03.2020 09:04 mostman077
A titration of vinegar with a solution of NaOH was performed. If 3.45 mL of vinegar needs 44.0 mL of 0.140 M NaOH to reach the equivalence point in a titration, Calculate the mass of acetic acid present in the vinegar sample: mastering chemistry answers

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2

Chemistry, 22.06.2019 17:30
The polymer used for the nonstick surface of cooking utensils is 24.0%c and 76%f by mass. what is the empirical formula of this polymer?
Answers: 2
You know the right answer?
A titration of vinegar with a solution of NaOH was performed. If 3.45 mL of vinegar needs 44.0 mL of...
Questions

Mathematics, 14.06.2020 10:57

Mathematics, 14.06.2020 10:57

Arts, 14.06.2020 10:57


Mathematics, 14.06.2020 10:57


Mathematics, 14.06.2020 10:57


Health, 14.06.2020 10:57


Mathematics, 14.06.2020 11:57

Spanish, 14.06.2020 11:57

Mathematics, 14.06.2020 11:57

Mathematics, 14.06.2020 11:57



Mathematics, 14.06.2020 11:57



Chemistry, 14.06.2020 11:57

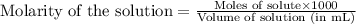
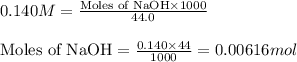
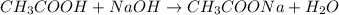
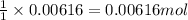 of acetic acid
of acetic acid