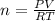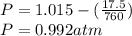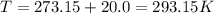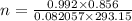Chemistry, 10.03.2020 09:05 lizzyhearts
Calculate the mass of O2 produced by the decomposition of KClO3 when 856 mL of O2 is collected over water at 20.0°C and 1.015 atm. (The vapor pressure of water at 20.0°C is 17.5 torr.)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:10
Amonoprotic acid is an acid that donates a single proton to the solution. suppose you have 0.140 g of a monoprotic acid dissolved in 35.0 ml of water. this solution is then neutralized with 14.5 ml of 0.110 m naoh. what is the molar mass of the acid?
Answers: 1

Chemistry, 22.06.2019 12:30
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1

Chemistry, 23.06.2019 01:00
Who examines and coordinates the cleanup of polluted sites?
Answers: 2

Chemistry, 23.06.2019 09:30
Where are the noble gases located in the periodic table? a. in the center b. on the left side c. in the upper right corner d. on the far right side
Answers: 1
You know the right answer?
Calculate the mass of O2 produced by the decomposition of KClO3 when 856 mL of O2 is collected over...
Questions



Physics, 29.07.2019 23:00







Computers and Technology, 29.07.2019 23:00

Mathematics, 29.07.2019 23:00

Mathematics, 29.07.2019 23:00

Mathematics, 29.07.2019 23:00


Biology, 29.07.2019 23:00

Mathematics, 29.07.2019 23:00


Business, 29.07.2019 23:00