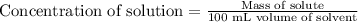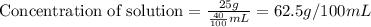Chemistry, 10.03.2020 11:28 jolleyrancher78
The concentration of a solution is equal to the mass of solute divided by the volume of solvent. Units of concentration are grams per 100 milliliters (g/100 mL, or g/dL). What is the concentration of this solution?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3

Chemistry, 22.06.2019 12:00
Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. a solution containing 3.50 g of sodium carbonate is mixed with one containing 5.00 g of silver nitrate. how many grams of sodium carbonate, silver nitrate, silver carbonate, and sodium nitrate are present after the reaction is complete?
Answers: 2

Chemistry, 22.06.2019 14:30
Amixture that has two or more substances that are spread out evenly is called a. compound b. heterogeneous c. substance d. homogeneous
Answers: 1

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2
You know the right answer?
The concentration of a solution is equal to the mass of solute divided by the volume of solvent. Uni...
Questions



Mathematics, 20.01.2021 01:00

Physics, 20.01.2021 01:00


Health, 20.01.2021 01:00

English, 20.01.2021 01:00

Mathematics, 20.01.2021 01:00



Chemistry, 20.01.2021 01:00

Mathematics, 20.01.2021 01:00


Mathematics, 20.01.2021 01:00

Mathematics, 20.01.2021 01:00


Mathematics, 20.01.2021 01:00

Computers and Technology, 20.01.2021 01:00

History, 20.01.2021 01:00