
Chemistry, 10.03.2020 08:21 lovebunny33921
CO(g)+2H2(g)⇌CH3OH(g)CO(g)+2H2(g)⇌C H3OH(g) This reaction is carried out at a different temperature with initial concentrations of [CO]=0.27M[CO]=0.27M and [H2]=0.49M[H2]=0.49M. At equilibrium, the concentration of CH3OHCH3OH is 0.11 MM. Find the equilibrium constant at this temperature.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 05:30
What happens to the atomic radius when an elctron is lost
Answers: 1

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2
You know the right answer?
CO(g)+2H2(g)⇌CH3OH(g)CO(g)+2H2(g)⇌C H3OH(g) This reaction is carried out at a different temperature...
Questions

Physics, 09.11.2020 09:50

Mathematics, 09.11.2020 09:50

Mathematics, 09.11.2020 09:50

Mathematics, 09.11.2020 09:50

Mathematics, 09.11.2020 09:50

Mathematics, 09.11.2020 09:50

English, 09.11.2020 09:50

History, 09.11.2020 09:50


Mathematics, 09.11.2020 09:50


Chemistry, 09.11.2020 09:50




Chemistry, 09.11.2020 09:50

English, 09.11.2020 09:50

Mathematics, 09.11.2020 09:50

English, 09.11.2020 09:50

History, 09.11.2020 09:50

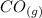 +
+ 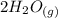 ⇄
⇄ 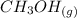
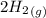 ⇄
⇄ ![K_C = \frac{[CH_3OH]}{[CO][H_2]^2}](/tpl/images/0540/6074/3667b.png)
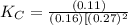
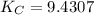
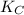 = 9.4
= 9.4

