Consider the following reaction, equilibrium concentrations, and equilibrium constant at
a par...

Chemistry, 10.03.2020 06:15 enevjordan
Consider the following reaction, equilibrium concentrations, and equilibrium constant at
a particular temperature. Determine the equilibrium concentration of H2O(g): [C2H4]eq = 0.015
M, [C2H5OH]eq = 1.69 M
C2H4(g) + H2O(g) ? C2H5OH(g) Kc = 9.0

Answers: 1
Another question on Chemistry


Chemistry, 21.06.2019 22:30
How many moles are in 250 grams of tungsten (w)? * 4.4x10^23 moles 4.2x10^23 moles 0.7 moles 1.4 moles
Answers: 3

Chemistry, 22.06.2019 18:00
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1

Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1
You know the right answer?
Questions

English, 05.11.2020 20:10


Mathematics, 05.11.2020 20:10

Mathematics, 05.11.2020 20:10


History, 05.11.2020 20:10


English, 05.11.2020 20:10


Chemistry, 05.11.2020 20:10

Mathematics, 05.11.2020 20:10

Social Studies, 05.11.2020 20:10

Mathematics, 05.11.2020 20:10



English, 05.11.2020 20:10

English, 05.11.2020 20:10


History, 05.11.2020 20:10

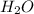 is 12.5 M
is 12.5 M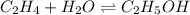
![K_{c}=\frac{[C_{2}H_{5}OH]}{[C_{2}H_{4}][H_{2}O]}](/tpl/images/0540/2224/b68d8.png)
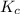 represents equilibrium constant in terms of concentration and species inside third bracket represent equilibrium concentrations
represents equilibrium constant in terms of concentration and species inside third bracket represent equilibrium concentrations![[C_{2}H_{4}]=0.015M](/tpl/images/0540/2224/3b394.png) ,
, ![[C_{2}H_{5}OH]=1.69M](/tpl/images/0540/2224/5d6f4.png) and
and 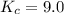
![[H_{2}O]=\frac{[C_{2}H_{5}OH]}{[C_{2}H_{4}]\times K_{c}}=\frac{1.69}{0.015\times 9.0}=12.5M](/tpl/images/0540/2224/0a407.png)


