
Chemistry, 10.03.2020 04:52 Katie123amazing
Calculate ΔH∘f for NO(g) at 435 K, assuming that the heat capacities of reactants and products are constant over the temperature interval at their values at 298.15 K. Molar heat capacities of NO(g), N2(g), and O2(g) at 298.15 K are 29.86, 29.13, and 29.38 J⋅K−1⋅mol−1. The standard enthalpy of formation of NO(g) is 91.3 kJ⋅mol−1 at 298.15 K.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2

Chemistry, 22.06.2019 07:20
Which of these conditions most likely produces an unstable isotope?
Answers: 1

Chemistry, 22.06.2019 12:30
Consider the four elements above. which one of these elements will combine with oxygen in a 1: 1 ratio?
Answers: 3
You know the right answer?
Calculate ΔH∘f for NO(g) at 435 K, assuming that the heat capacities of reactants and products are c...
Questions


Social Studies, 01.10.2019 00:30


Chemistry, 01.10.2019 00:30

Biology, 01.10.2019 00:30


Social Studies, 01.10.2019 00:30

English, 01.10.2019 00:30


Social Studies, 01.10.2019 00:30


Mathematics, 01.10.2019 00:30


History, 01.10.2019 00:30

Physics, 01.10.2019 00:30




Social Studies, 01.10.2019 00:30

Mathematics, 01.10.2019 00:30

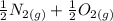 ------>
------>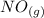
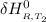 =
= 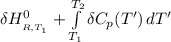
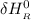 = enthalpy of reaction
= enthalpy of reaction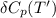 = the difference in the heat capacities of the products and the reactants.
= the difference in the heat capacities of the products and the reactants.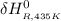 =
= 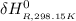

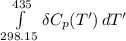
![1(91300 J.mol^{-1} ) +\int\limits^{435}_{298.15} [{(29.86)-\frac{1}{2}(29.38)-\frac{1}{2}29.13}]J.K^{-1}.mol^{-1} \, dT'](/tpl/images/0540/0534/3b971.png)


