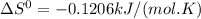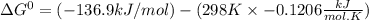Chemistry, 10.03.2020 03:39 puchie1225
The following reaction has the thermodynamic values at 298 K: ΔH° = -136.9 kJ/mol and ΔS° = -120.6 J/mol K. Calculate ΔG° at 298 K for this reaction in kJ/mol (Enter your answer to four significant figures.):

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:50
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

You know the right answer?
The following reaction has the thermodynamic values at 298 K: ΔH° = -136.9 kJ/mol and ΔS° = -120.6 J...
Questions

Computers and Technology, 28.09.2019 22:30

Chemistry, 28.09.2019 22:30

Chemistry, 28.09.2019 22:30

Geography, 28.09.2019 22:30

Mathematics, 28.09.2019 22:30

English, 28.09.2019 22:30




Mathematics, 28.09.2019 22:30





History, 28.09.2019 22:30


English, 28.09.2019 22:30

Health, 28.09.2019 22:30


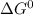 at 298 K is -101.0 kJ/mol
at 298 K is -101.0 kJ/mol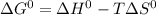
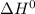 , T and
, T and 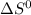 represent change in free energy in standard state, change in enthalpy in standard state, temperature in kelvin scale and change in entropy in standard state.
represent change in free energy in standard state, change in enthalpy in standard state, temperature in kelvin scale and change in entropy in standard state.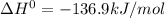 , T = 298 K and
, T = 298 K and