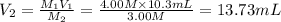Chemistry, 10.03.2020 01:07 jocelyngracia
A chemist must dilute 10.3mL of 4.00M aqueous sodium chloride NaCl solution until the concentration falls to 3.00M . He'll do this by adding distilled water to the solution until it reaches a certain final volume.
Required:
Calculate this final volume, in liters.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1

Chemistry, 23.06.2019 05:30
Elizabeth has two separate samples of the same substance. sample is in the liquid state, and the other is in the solid state. the two samples most likely differ in which property?
Answers: 1

Chemistry, 23.06.2019 06:30
Achemist is studying the following equilibirum, which has the given equilibrium constant at a certain temperature: 2 no(g) + cl2(g) < => 2 nocl(g) kp = 2 x 10^(-6)he fills a reaction vessel at this temperature with 13. atm of nitrogen monoxide gas and 12. atm of chlorine gas. use this data to answer the questions: a. can you predict the equilibrium pressure of noci, using only the tools available to you within aleks? y/nb. if you said yes, then enter the equilibrium pressure of nocl at right. round your answer to 1 significant digit.
Answers: 1
You know the right answer?
A chemist must dilute 10.3mL of 4.00M aqueous sodium chloride NaCl solution until the concentration...
Questions



History, 23.06.2021 06:30


Mathematics, 23.06.2021 06:30

Social Studies, 23.06.2021 06:30


English, 23.06.2021 06:30




Mathematics, 23.06.2021 06:30




Mathematics, 23.06.2021 06:30


Social Studies, 23.06.2021 06:30


Physics, 23.06.2021 06:30

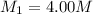
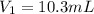
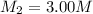
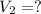
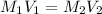 (dilution)
(dilution)