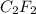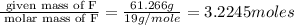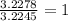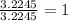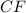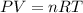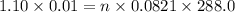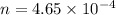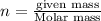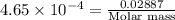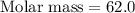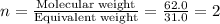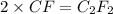Chemistry, 10.03.2020 00:29 jjimenez0276
A 0.02887 g sample of gas occupies 10.0 mL at 288.0 K and 1.10 atm. Upon further analysis, the compound is found to be 38.734 % C and 61.266 % F . What is the molecular formula of the compound?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
Select each correct answer. more than one answer may be correct. which of the following is a characteristic of unicellular organisms? they can possess tissues and organs. all of their functions are performed by a single cell. they are usually microscopic. each of their cells is specialized to perform a specific function.
Answers: 1

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 23.06.2019 03:30
In chemistry, the type of an atom (what element it is) is determined by: a) the number of protons it contains in its nucleus.b) the number of neutrons it contains in its nucleus.c) the number of protons it has in a cloud around the nucleus.d) the number of neutrons it has in a cloud around the nucleus.e) the number of electrons it exchanges with its neighbors.
Answers: 1

Chemistry, 23.06.2019 11:30
Jenny places a strip of ph paper into a solution. when she removes the ph paper, it has turned yellow-green. what should jenny do next to determine the ph of her solution? a. use a different testing method because the ph paper should not change colors b. place the ph paper into a machine that reads the ph of the solution c. compare the ph paper's color with the color of ph paper from another solution d. compare the ph paper's color with a chart of colors and ph ranges
Answers: 1
You know the right answer?
A 0.02887 g sample of gas occupies 10.0 mL at 288.0 K and 1.10 atm. Upon further analysis, the compo...
Questions


Mathematics, 07.10.2020 20:01

Biology, 07.10.2020 20:01


Mathematics, 07.10.2020 20:01




Physics, 07.10.2020 20:01




Arts, 07.10.2020 20:01

Mathematics, 07.10.2020 20:01


Mathematics, 07.10.2020 20:01

English, 07.10.2020 20:01

Social Studies, 07.10.2020 20:01


Mathematics, 07.10.2020 20:01