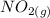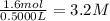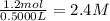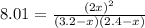Chemistry, 10.03.2020 00:27 TabbyKun00
Suppose a 500.mL flask is filled with 1.6mol of NO3 and 1.2mol of NO2 . The following reaction becomes possible: +NO3gNOg 2NO2g The equilibrium constant K for this reaction is 8.01 at the temperature of the flask. Calculate the equilibrium molarity of NO3 . Round your answer to two decimal places.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:30
In pea plants, the allele for tallness (t) is dominant to the allele for shortness (t). in the cross between a tall pea plant and a short pea plant shown below, what is the probability that the resulting offspring will be tall? whats the percent
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 22:30
The vapor pressure of ethanol is 1.00 × 102 mmhg at 34.90°c. what is its vapor pressure at 61.61°c? (δhvap for ethanol is 39.3 kj/mol.)
Answers: 2

Chemistry, 23.06.2019 18:20
Ahydrogen electron is elevated from level 1 to level 2. another electron is elevated from level 2 to level 4. the transition requiring the greatest energy change is?
Answers: 1
You know the right answer?
Suppose a 500.mL flask is filled with 1.6mol of NO3 and 1.2mol of NO2 . The following reaction becom...
Questions

Mathematics, 21.06.2019 15:30

Mathematics, 21.06.2019 15:30

Mathematics, 21.06.2019 15:30




English, 21.06.2019 15:30

Computers and Technology, 21.06.2019 15:30




English, 21.06.2019 15:30

History, 21.06.2019 15:30


Physics, 21.06.2019 15:30




Biology, 21.06.2019 15:30

Mathematics, 21.06.2019 15:30

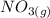 is 1.60 M
is 1.60 M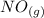 ⇄2
⇄2