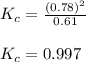Chemistry, 09.03.2020 23:59 pollywallythecat
Exactly 1.0 mol N2O4 is placed in an empty 1.0-L container and is allowed to reach equilibrium described by the equation N2O4(g) 2NO2(g) If at equilibrium the N2O4 is 39% dissociated, what is the value of the equilibrium constant, Kc, for the reaction under these conditions

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:30
If the element whose electric configuration ends in the d sublevel, the element is calssified as? a.inner transition b.noble gases c.representative d. transition
Answers: 2

Chemistry, 22.06.2019 13:50
Amap that uses a range of colors and shading to represent the elevation, depth, or landscape of specific features on earth is a/an map.
Answers: 3

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

Chemistry, 22.06.2019 21:40
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2
You know the right answer?
Exactly 1.0 mol N2O4 is placed in an empty 1.0-L container and is allowed to reach equilibrium descr...
Questions

Computers and Technology, 06.05.2021 19:40


Engineering, 06.05.2021 19:40


Mathematics, 06.05.2021 19:40

Medicine, 06.05.2021 19:40



Computers and Technology, 06.05.2021 19:40







Mathematics, 06.05.2021 19:40



Health, 06.05.2021 19:40

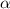 = 0.39
= 0.39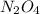 , c =
, c = 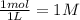
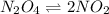
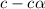
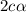
![N_2O_4=c-c\alpha =[1-(1\times 0.39)]=0.61M](/tpl/images/0539/1468/f8a46.png)
![NO_2=2c\alpha =[2\times 1\times 0.39]=0.78M](/tpl/images/0539/1468/5fcc4.png)
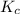 for above equation follows:
for above equation follows:![K_{c}=\frac{[NO_2]^2}{[N_2O_4]}](/tpl/images/0539/1468/1a559.png)