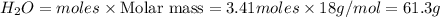Chemistry, 09.03.2020 23:57 jaynissiagreen16
The Ostwald process is used commercially to produce nitric acid, which is, in turn, used in many modern chemical processes. In the first step of the Ostwald process, ammonia is reacted with oxygen gas to produce nitric oxide and water. What is the maximum mass of H 2 O that can be produced by combining 91.0 g of each reactant? 4 NH 3 ( g ) + 5 O 2 ( g ) ⟶ 4 NO ( g ) + 6 H 2 O ( g )

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:30
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible?
Answers: 2

Chemistry, 21.06.2019 20:30
Which sentence best describes the formation of igneous rock? a- lava on the surface dries up and makes arock b_melted rocks cools and forms crystals c_rocks under tremendous heat and pressure d_magma is melted rock underground
Answers: 1

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

Chemistry, 22.06.2019 22:00
8) warming your hands by a fire is an example if which heat transfer? a. conduction b. convection c. radiation d. none of these
Answers: 1
You know the right answer?
The Ostwald process is used commercially to produce nitric acid, which is, in turn, used in many mod...
Questions

Mathematics, 22.10.2019 17:50


Health, 22.10.2019 17:50


Mathematics, 22.10.2019 17:50

Chemistry, 22.10.2019 17:50

History, 22.10.2019 17:50

Chemistry, 22.10.2019 17:50

Biology, 22.10.2019 17:50

Mathematics, 22.10.2019 17:50






Business, 22.10.2019 17:50




History, 22.10.2019 17:50

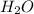 will be produced from the given masses of both reactants.
will be produced from the given masses of both reactants.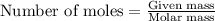
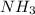
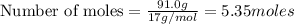
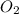
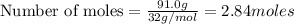
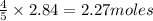 of
of 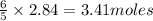 of
of