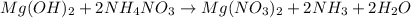Chemistry, 09.03.2020 23:56 SushiMagic
When ammonium nitrate is added to a suspension of magnesium hydroxide in water, the Mg(OH)2 dissolves. Write a net ionic equation to show how this occurs. Do not include physical states and use the smallest possible integer coefficients.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:50
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1

Chemistry, 22.06.2019 14:00
Which of the following is true about a carbonated soft drink? . the carbon dioxide is the solvent, and water is the solute.. the water is the solution, and carbon dioxide is the solvent.. the carbon dioxide is the solution, and the water is the solvent.. the water is the solvent, and the carbon dioxide is the solute.. .
Answers: 1

Chemistry, 22.06.2019 22:30
The vapor pressure of ethanol is 1.00 × 102 mmhg at 34.90°c. what is its vapor pressure at 61.61°c? (δhvap for ethanol is 39.3 kj/mol.)
Answers: 2

Chemistry, 23.06.2019 03:30
In chemistry, the type of an atom (what element it is) is determined by: a) the number of protons it contains in its nucleus.b) the number of neutrons it contains in its nucleus.c) the number of protons it has in a cloud around the nucleus.d) the number of neutrons it has in a cloud around the nucleus.e) the number of electrons it exchanges with its neighbors.
Answers: 1
You know the right answer?
When ammonium nitrate is added to a suspension of magnesium hydroxide in water, the Mg(OH)2 dissolve...
Questions

History, 22.10.2019 10:50



Chemistry, 22.10.2019 10:50


Physics, 22.10.2019 10:50




History, 22.10.2019 10:50


English, 22.10.2019 10:50




Social Studies, 22.10.2019 10:50

History, 22.10.2019 10:50

World Languages, 22.10.2019 11:00

Social Studies, 22.10.2019 11:00

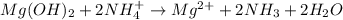
 in
in  reacts with
reacts with 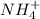 to form
to form 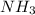 and
and 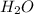 .
.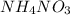 is added to suspension of
is added to suspension of