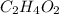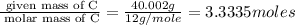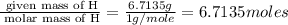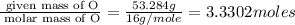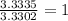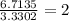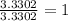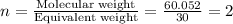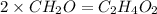Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:00
What effect does a decrease in temperature have on the overall rate of a chemical reaction? a decrease in temperature decreases . the reaction rate will
Answers: 1

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1


Chemistry, 22.06.2019 15:00
How is the shape of the poem “peer” connected to its meaning?
Answers: 2
You know the right answer?
The percent composition by mass of an unknown compound with a molecular mass of 60.052 amu is 40.002...
Questions



Mathematics, 15.10.2019 19:30

Mathematics, 15.10.2019 19:30



Mathematics, 15.10.2019 19:30





Computers and Technology, 15.10.2019 19:30


Mathematics, 15.10.2019 19:30

Biology, 15.10.2019 19:30



Biology, 15.10.2019 19:30

Social Studies, 15.10.2019 19:30

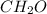 and
and