
Chemistry, 09.03.2020 02:49 connermichaela
A cyclist rides at the rate of 1cm per second how many miles will the cyclist travel in a year?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
What three natural resources are found in the great lakes region
Answers: 2

Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Chemistry, 23.06.2019 00:30
The footprints of a dinosaur and the burrow of an ancient shrimp are examples of which kind of fossils
Answers: 2

Chemistry, 23.06.2019 05:30
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table? atom p has an estimated zeff of 7 and is therefore to the left of atom q, which has a zeff of 6. atom p has an estimated zeff of 7 and is therefore to the right of atom q, which has a zeff of 6. atom p has an estimated zeff of 5 and is therefore below atom q, which has a zeff of 4. atom p has an estimated zeff of 5 and is therefore above atom q, which has a zeff of 4.
Answers: 3
You know the right answer?
A cyclist rides at the rate of 1cm per second how many miles will the cyclist travel in a year?...
Questions



Biology, 13.10.2019 23:30

English, 13.10.2019 23:30

History, 13.10.2019 23:30


Biology, 13.10.2019 23:30



Biology, 13.10.2019 23:30




Biology, 13.10.2019 23:30


Biology, 13.10.2019 23:30


Arts, 13.10.2019 23:30

Chemistry, 13.10.2019 23:30

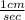 ×
× 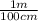 =
= 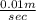
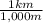 =
=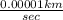
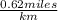 =
= 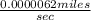
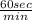 =
= 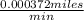
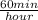 =
= 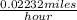
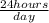 =
= 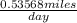
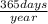 =
= 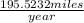 or 195.5232
or 195.5232 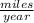 = answer
= answer


