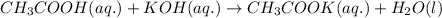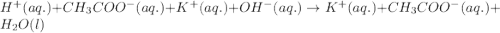Chemistry, 07.03.2020 04:57 BlehBlehBlehBleh
Equal volumes of 0.25 M acetic acid and 0.25 M potassium hydroxide are combined. Write the net ionic equation for the reaction and identify the aqueous species that have the highest concentrations at equilibrium. Justify your answer.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

Chemistry, 22.06.2019 02:10
3.) for each of the following compounds, draw the major organic product of reaction with hcl or naoh and circle whether the starting materials and products will be more soluble in organic solvent or water benzoic acid + hcl: benzoic acid + naoh: oh benzoic acid water/organic water organic fluorenone hс: fluorenone + naoh: fluorenone water/organic water/organic веnzocaine + hci: benzocaine + n»oh: h2n benzocaine water/organic water organic o=
Answers: 3

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2
You know the right answer?
Equal volumes of 0.25 M acetic acid and 0.25 M potassium hydroxide are combined. Write the net ionic...
Questions

Mathematics, 10.04.2020 06:00


Mathematics, 10.04.2020 06:00

Mathematics, 10.04.2020 06:00


Mathematics, 10.04.2020 06:00

Mathematics, 10.04.2020 06:00






English, 10.04.2020 06:00



Mathematics, 10.04.2020 06:00

Mathematics, 10.04.2020 06:00

Mathematics, 10.04.2020 06:00

Business, 10.04.2020 06:00

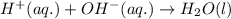 and all the ions have equal concentrations at equilibrium
and all the ions have equal concentrations at equilibrium