
Chemistry, 07.03.2020 04:14 mckleinrivero
The rate constant for a certain reaction is kkk = 3.50×10−3 s−1s−1 . If the initial reactant concentration was 0.450 MM, what will the concentration be after 19.0 minutes?
A zero-order reaction has a constant rate of 3.50×10−4 M/sM/s. If after 65.0 seconds the concentration has dropped to 3.50×10−2 MM, what was the initial concentration?

Answers: 2
Another question on Chemistry


Chemistry, 21.06.2019 23:50
How does a scientist the government? a. the scientist tells people in society what to do. b. the scientist determines the policies that the government spends money on. c. the scientist provides unbiased information to the government. d. the scientist makes laws based on his or her research results.
Answers: 1

Chemistry, 22.06.2019 03:00
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
You know the right answer?
The rate constant for a certain reaction is kkk = 3.50×10−3 s−1s−1 . If the initial reactant concent...
Questions









Biology, 17.05.2021 17:00


Mathematics, 17.05.2021 17:00

Mathematics, 17.05.2021 17:00

Mathematics, 17.05.2021 17:00

Physics, 17.05.2021 17:00

Mathematics, 17.05.2021 17:00





French, 17.05.2021 17:00

![Rate = k[A][B]](/tpl/images/0537/5969/ab111.png) . As the data are given in the question, the rate constant is
. As the data are given in the question, the rate constant is 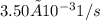 , the initial concentration
, the initial concentration![[A]_o = 0.450 M](/tpl/images/0537/5969/1d14c.png) and time is 19 minutes that is the 1140s.
and time is 19 minutes that is the 1140s.
![ln[A] = ln[A]_o - kt\\ln[A] = ln (0.450) - (3.50*10^-^3)1140\\ln[A] = -0.799 - 3.99\\ln[A] = -4.789\\[A] = 0.0083M](/tpl/images/0537/5969/91bfd.png)
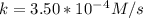 Time = 65 s
Final Concentration [A] =
Time = 65 s
Final Concentration [A] = 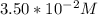
![[A] = [A]_o - kt[A]_o = [A] + kt\\[A]o = 3.50*10^{-2} + (3.50*10^{-4}) * 65\\[A]_o = 3.50*10^{-2} + 0.02275\\[A]_o = 0.05775 M](/tpl/images/0537/5969/3e5cb.png)


