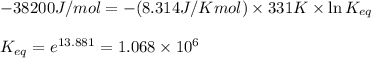Chemistry, 07.03.2020 03:46 Joshuafranklindude
In the Mond process for the purification of nickel, carbon monoxide is reacted with heated nickel to produce Ni(CO)4, which is a gas and can therefore be separated from solid impurities: Ni(s) + 4CO(g) ⇌ Ni(CO)4(g) Given that the standard free energies of formation of CO(g) and Ni(CO)4(g) are −137.3 and −587.4 kJ/mol, respectively, calculate the equilibrium constant of the reaction at 58.0°C. Assume that ΔG o f is temperature-independent.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Which answer lists the fundamental forces in order from strongest to weakest
Answers: 1

Chemistry, 22.06.2019 00:20
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2

Chemistry, 22.06.2019 06:00
In an investigation that uses the scientific method, which step immediately follows making a hypothesis? o summarizing the results o asking a question o making observations designing an experiment mark this and retum save and exit next submit
Answers: 2

Chemistry, 22.06.2019 07:00
Which set of characteristics best describes igneous rock? a) largest type of rock, made of organic matter, hardest type of rock b) least abundant type of rock, made of other rocks, made mostly of minerals c) found on all continents, contains wavy bands of stripes, contains fossils d) most abundant type in earth's crust, made of magma/lava, contains no fossils
Answers: 1
You know the right answer?
In the Mond process for the purification of nickel, carbon monoxide is reacted with heated nickel to...
Questions







Biology, 23.06.2019 11:30

Chemistry, 23.06.2019 11:30

Mathematics, 23.06.2019 11:30

Mathematics, 23.06.2019 11:30

History, 23.06.2019 11:30

Mathematics, 23.06.2019 11:30



Chemistry, 23.06.2019 11:30


Mathematics, 23.06.2019 11:30


Mathematics, 23.06.2019 11:30

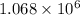
![\Delta G^o_{rxn}=\sum [n\times \Delta G^o_{(product)}]-\sum [n\times \Delta G^o_{(reactant)}]](/tpl/images/0537/4791/f0852.png)
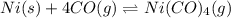
![\Delta G^o_{rxn}=[(1\times \Delta G^o_{(Ni(CO)_4(g))})]-[(1\times \Delta G^o_{(Ni(s))})+(4\times \Delta G^o_{(CO(g))})]](/tpl/images/0537/4791/5f698.png)
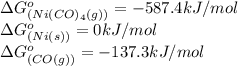
![\Delta G^o_{rxn}=[(1\times (-587.4))]-[(1\times (0))+(4\times (-137.3))]\\\\\Delta G^o_{rxn}=-38.2kJ/mol](/tpl/images/0537/4791/37589.png)
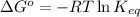
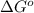 = Standard Gibbs free energy = -38.2 kJ/mol = -38200 J/mol (Conversion factor: 1 kJ = 1000 J )
= Standard Gibbs free energy = -38.2 kJ/mol = -38200 J/mol (Conversion factor: 1 kJ = 1000 J )![58^oC=[273+58]K=331K](/tpl/images/0537/4791/2a33f.png)
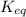 = equilibrium constant at 58°C = ?
= equilibrium constant at 58°C = ?