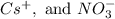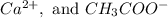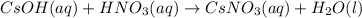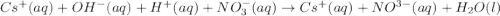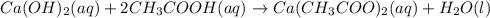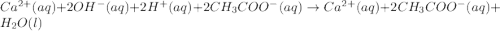In the following acid-base reactions, identify the spectator ions, if any:
(i) Cesium hydroxide(aq) + nitric acid(aq) =>
(ii) Calcium hydroxide(aq) + acetic acid(aq) =>
a.
(i) Cs+ and NO3-
(ii) no spectator ions
b.
(i) Cs+ and NO3-
(ii) H+ and OH-
c.
(i) no spectator ions
(ii) H+ and OH-
d.
(i) Cs+ and NO3-
(ii) Ca2+ and acetate ion.
e.
(i) no spectator ions
(ii) no spectator ions

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 12:00
Under normal conditions, describe how increasing the temperatures effects the solubility of a typical salt
Answers: 1

You know the right answer?
In the following acid-base reactions, identify the spectator ions, if any:
(i) Cesium hydroxid...
(i) Cesium hydroxid...
Questions


English, 10.12.2021 19:30

Mathematics, 10.12.2021 19:30





Mathematics, 10.12.2021 19:30




History, 10.12.2021 19:30


English, 10.12.2021 19:30

Mathematics, 10.12.2021 19:30

Spanish, 10.12.2021 19:30

Arts, 10.12.2021 19:30

Spanish, 10.12.2021 19:30