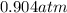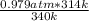Chemistry, 06.03.2020 23:49 RoyalGurl01
A sealed container holding 0.0255 L of an ideal gas at 0.979 atm and 67 ∘ C is placed into a refrigerator and cooled to 41 ∘ C with no change in volume. Calculate the final pressure of the gas. P = atm

Answers: 2
Another question on Chemistry

Chemistry, 23.06.2019 02:10
Detrimental the length of the object shown 1. 97.8mm 2. 97.80 mm 3. 97mm 4. 98mm
Answers: 2

Chemistry, 23.06.2019 10:00
An uncovered pot of water lies out in the sun. which statements correctly describe what happens at the surface of the liquid water? 1. the vapor pressure remains constant regardless of the water temperature. 2. the vapor pressure is produced by water molecules that have evaporated. 3. the vapor pressure increases as the sun heats the water in the pot. 4. evaporation stops once the vapor pressure reaches a certain point. 5. evaporation and condensation both occur on the liquid’s surface.
Answers: 3

Chemistry, 23.06.2019 12:00
Which of the following statements is true? a. most heat energy is easily recovered and used for useful actions. b. friction causes molecules to vibrate more slowly. burning air and gasoline in an c. engine changes chemical energy into mechanical energy. it is impossible to d. change mechanical energy into mechanical energy.
Answers: 1

Chemistry, 23.06.2019 14:00
How is the electron sea model of metallic bonding different from the band theory? how are they the same? give at least one similarity and one difference between the models
Answers: 2
You know the right answer?
A sealed container holding 0.0255 L of an ideal gas at 0.979 atm and 67 ∘ C is placed into a refrige...
Questions






Social Studies, 16.06.2021 17:10

Mathematics, 16.06.2021 17:10


Mathematics, 16.06.2021 17:10

Social Studies, 16.06.2021 17:10

History, 16.06.2021 17:10

Social Studies, 16.06.2021 17:10




English, 16.06.2021 17:10




Chemistry, 16.06.2021 17:10