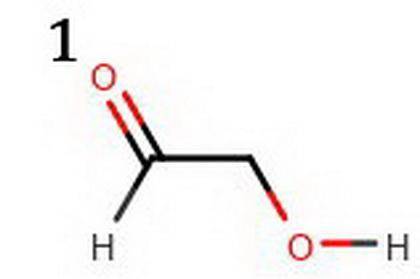
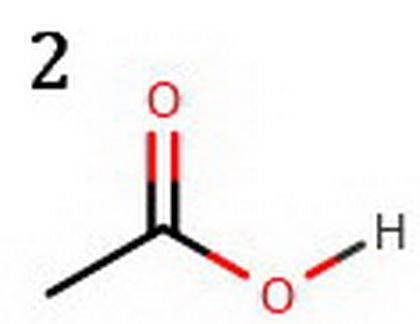
The following two compounds are constitutional isomers. Identify which of these is expected to be more acidic, and explain your choice. a. The compound below is more acidic because its conjugate base is more resonance stabilized. b. The conjugate base of the other compound is not as much resonance stabilized. c. The compound below is more acidic because its conjugate base is more resonance stabilized. d. The conjugate base of the other compound is not as much resonance stabilized. e. The compound below is more acidic because its conjugate base is resonance stabilized. f. The conjugate base of the other compound is not resonance stabilized. Tg. he compound below is more acidic because its conjugate base is resonance stabilized. h. The conjugate base of the other compound is not resonance stabilized.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:30
Compare and contrast refraction of light and sound will give brainliest
Answers: 1

Chemistry, 23.06.2019 09:10
In a 28 g serving of cheese curls there are 247mg of sodium. how much sodium is in a 12.5 ounce bag
Answers: 1

Chemistry, 23.06.2019 13:20
In the haber reaction, patented by german chemist fritz haber in 1908, dinitrogen gas combines with dihydrogen gas to produce gaseous ammonia. this reaction is now the first step taken to make most of the world's fertilizer. suppose a chemical engineer studying a new catalyst for the haber reaction finds that 671 liters per second of dinitrogen are consumed when the reaction is run at 271c and 0.99atm. calculate the rate at which ammonia is being produced. give your answer in kilograms per second. round your answer to significant digits.
Answers: 3

Chemistry, 23.06.2019 19:10
An engineer that designs buildings is a(n) a) civil engineer b) mechanical engineer c) chemical engineer d) aerospace engineer
Answers: 2
You know the right answer?
The following two compounds are constitutional isomers. Identify which of these is expected to be mo...
Questions

English, 22.04.2020 21:53


Computers and Technology, 22.04.2020 21:53

Biology, 22.04.2020 21:53

Mathematics, 22.04.2020 21:54

Chemistry, 22.04.2020 21:54

Mathematics, 22.04.2020 21:54



Advanced Placement (AP), 22.04.2020 21:54

Mathematics, 22.04.2020 21:54


Biology, 22.04.2020 21:54


English, 22.04.2020 21:54

History, 22.04.2020 21:54



Mathematics, 22.04.2020 21:54

Computers and Technology, 22.04.2020 21:54