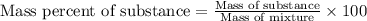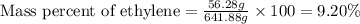Ethylene () is the starting point for a wide array of industrial chemical syntheses. For example, worldwide about of polyethylene are made from ethylene each year, for use in everything from household plumbing to artificial joints. Natural sources of ethylene are entirely inadequate to meet world demand, so ethane () from natural gas is "cracked" in refineries at high temperature in a kinetically complex reaction that produces ethylene gas and hydrogen gas. Suppose an engineer studying ethane cracking fills a reaction tank with of ethane gas and raises the temperature to . He believes at this temperature. Calculate the percent by mass of ethylene the engineer expects to find in the equilibrium gas mixture. Round your answer to significant digits. Note for advanced students: the engineer may be mistaken about the correct value of , and the mass percent of ethylene you calculate may not be what he actually observes.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 16:30
An atom with 7 protons, 6 neutrons, and 7 electrons has an atomic mass of amu. (enter a whole number.) numerical answers expected! answer for blank 1:
Answers: 3
You know the right answer?
Ethylene () is the starting point for a wide array of industrial chemical syntheses. For example, wo...
Questions

Mathematics, 19.11.2020 06:30


English, 19.11.2020 06:30

Advanced Placement (AP), 19.11.2020 06:30

Arts, 19.11.2020 06:30

Biology, 19.11.2020 06:30

Chemistry, 19.11.2020 06:30



Computers and Technology, 19.11.2020 06:30

Mathematics, 19.11.2020 06:30

English, 19.11.2020 06:30

English, 19.11.2020 06:30

English, 19.11.2020 06:30


Mathematics, 19.11.2020 06:30

Mathematics, 19.11.2020 06:30



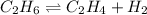
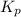 for above equation follows:
for above equation follows: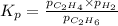
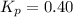
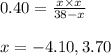
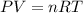 ..........(1)
..........(1)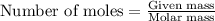 .....(2)
.....(2)![P=34.3atm\\V=30.0L\\R=0.0821\text{ L atm }mol^{-1}K^{-1}\\T=400^oC=[400+273]=673K](/tpl/images/0534/8160/3194c.png)

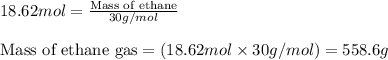
![P=3.70atm\\V=30.0L\\R=0.0821\text{ L atm }mol^{-1}K^{-1}\\T=400^oC=[400+273]=673K](/tpl/images/0534/8160/4b69b.png)