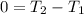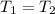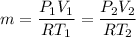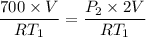Chemistry, 05.03.2020 22:37 gibsonchristian96
An insulated rigid tank is divided into two equal parts by a partition. Initially, one part contains 4 kg of an ideal gas at 700 kPa and 59°C, and the other part is evacuated. The partition is now removed, and the gas expands into the entire tank. Determine the final temperature and pressure in the tank.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 2

Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3

Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1
You know the right answer?
An insulated rigid tank is divided into two equal parts by a partition. Initially, one part contains...
Questions

Chemistry, 11.10.2019 19:00





Social Studies, 11.10.2019 19:00


Mathematics, 11.10.2019 19:00




Physics, 11.10.2019 19:00



Mathematics, 11.10.2019 19:00

Computers and Technology, 11.10.2019 19:00




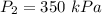
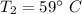
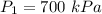
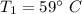
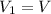

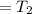
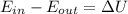
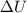 = Change in the internal energy of the gas
= Change in the internal energy of the gas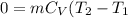 ) ( Cv=Specific heat capacity at constant volume)
) ( Cv=Specific heat capacity at constant volume)