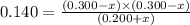Consider mixture C, which will cause the net reaction to proceed in reverse. Concentration (M)initial:change:equilibrium:[XY]0 .200+x0.200+x←net⇌[X]0.300−x0.300−x +[Y]0.300−x0.300−x The change in concentration, x, is positive for the reactants because they are produced and negative for the products because they are consumed.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
In which layer of earth do most earthauakes occur a_ inner core b_outer core c_mantle d_crust
Answers: 1

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 13:00
What happens to the average kinetic energy of a gas when the particles of the gas collide against each other at a constant temperature and volume? explain your answer.
Answers: 3

Chemistry, 23.06.2019 08:00
Which of the following notations would be the appropriate and final way to display the formula for magnesium chloride a. mgcl2 b. mg+2cl–1 c. mgcl2 d. mgcl
Answers: 2
You know the right answer?
Consider mixture C, which will cause the net reaction to proceed in reverse. Concentration (M)initia...
Questions

Mathematics, 07.05.2021 23:30


Engineering, 07.05.2021 23:30

Mathematics, 07.05.2021 23:30




Mathematics, 07.05.2021 23:30


Computers and Technology, 07.05.2021 23:30

Mathematics, 07.05.2021 23:30

English, 07.05.2021 23:30




Health, 07.05.2021 23:30

Mathematics, 07.05.2021 23:30



Biology, 07.05.2021 23:30

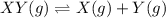
![K_c=\farc{[X][Y]}{[XY]}](/tpl/images/0534/4698/91cd9.png)