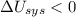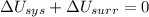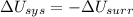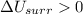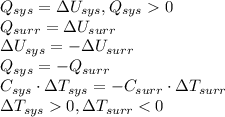Chemistry, 04.03.2020 23:29 isaiahcannon5709
Determine if each of the statements is True or False regarding the First Law of Thermodynamics.
1. If the system loses energy to the surroundings, the surroundings could also lose energy to the system.
2. If the system gains thermal energy from the surroundings, the temperature of the surroundings decreases.
3 If the system gains 25 kJ of energy from the surroundings without doing any work on the surroundings, the surroundings could lose 20 kJ of energy.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change. when the temperature in a room increases from 25°c to 33°c, changes from a solid to a liquid. in a lab, methane and nitrogen are cooled from -170°c to -200°c. the methane freezes and the nitrogen . when gold is heated to 2,856°c it changes from a liquid to a .
Answers: 2

Chemistry, 21.06.2019 18:30
Calculate the change in entropy if br2(l) is converted to br2(g). s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 3

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

You know the right answer?
Determine if each of the statements is True or False regarding the First Law of Thermodynamics.
Questions



Mathematics, 10.03.2020 19:07


English, 10.03.2020 19:07

Mathematics, 10.03.2020 19:07


Physics, 10.03.2020 19:07




Computers and Technology, 10.03.2020 19:07