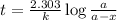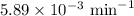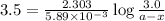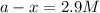Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2


Chemistry, 22.06.2019 19:30
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2
You know the right answer?
N2O5 decomposes to form NO2 and O2 with first-order kinetics. The initial concentration of N2O5 is 3...
Questions






History, 14.11.2019 13:31

Health, 14.11.2019 13:31


Social Studies, 14.11.2019 13:31

Mathematics, 14.11.2019 13:31


Social Studies, 14.11.2019 13:31


History, 14.11.2019 13:31

English, 14.11.2019 13:31




Biology, 14.11.2019 13:31

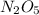 is, 2.9 M
is, 2.9 M