
Chemistry, 04.03.2020 23:43 angel213326
Magnesium (used in the manufacture of light alloys) reacts with iron(III) chloride to form magnesium chloride and iron. A mixture of 41.0 g of magnesium and 175.0 g of iron(III) chloride is allowed to react. Identify the limiting reactant and determine the mass of the excess reactant present in the vessel when the reaction is complete.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which of the dna typing techniques do you think you would choose if you had to analyze a dna sample? why?
Answers: 1

Chemistry, 22.06.2019 08:20
What is the formula for the compound dinitrogen pentoxide? a. n4o5 b. n5o4 c. n4o6 d. n5o2 e. n2o5
Answers: 3

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1

Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2
You know the right answer?
Magnesium (used in the manufacture of light alloys) reacts with iron(III) chloride to form magnesium...
Questions


Biology, 25.11.2021 06:20





Mathematics, 25.11.2021 06:20

English, 25.11.2021 06:20


English, 25.11.2021 06:20


Chemistry, 25.11.2021 06:20

Spanish, 25.11.2021 06:20



Social Studies, 25.11.2021 06:20



Social Studies, 25.11.2021 06:20

Biology, 25.11.2021 06:20

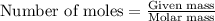 .....(1)
.....(1)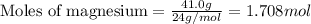
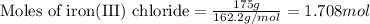
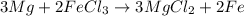
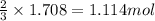 of iron(III) chloride
of iron(III) chloride


