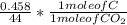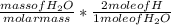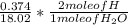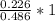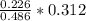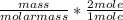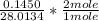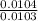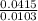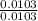Chemistry, 05.03.2020 00:53 hbhdnjdndj1867
Dimethylhydrazine is a carbon-hydrogen-nitrogen compound used in rocket fuels. When burned in an excess of oxygen, a 0.312 gg sample yields 0.458 gg CO2CO2 and 0.374 gg H2OH2O. The nitrogen content of a 0.486 gg sample is converted to 0.226 gg N2N2.What is the empirical formula of dimethylhydrazine?Express your answer as a chemical formula.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2

Chemistry, 23.06.2019 04:00
Calculate the mass of 0.750 mol of the following substance. na3po4. , i'm not quite sure on how to set up the problem to solve! : (
Answers: 1

Chemistry, 23.06.2019 09:00
How many moles of sulfur dioxide are in 2.26 × 10^33 sulfur dioxide molecules?
Answers: 3
You know the right answer?
Dimethylhydrazine is a carbon-hydrogen-nitrogen compound used in rocket fuels. When burned in an exc...
Questions

History, 14.07.2020 20:01





Mathematics, 14.07.2020 20:01

Mathematics, 14.07.2020 20:01

Physics, 14.07.2020 20:01


Mathematics, 14.07.2020 20:01


Mathematics, 14.07.2020 20:01



English, 14.07.2020 20:01

Geography, 14.07.2020 20:01

History, 14.07.2020 20:01

Mathematics, 14.07.2020 20:01

Mathematics, 14.07.2020 20:01