Chemistry, 04.03.2020 19:17 lberries08
Uppose you perform a titration of an unknown weak acid solution. You start with 4.00 mL of the weak acid and find that it takes 12.8 mL of 0.0500 M NaOH to reach the equivalence point. What is the concentration of the unknown weak acid solution?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1


Chemistry, 22.06.2019 12:20
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1

Chemistry, 23.06.2019 03:00
Describe the properties of sodium, chlorine, and sodium chloride
Answers: 1
You know the right answer?
Uppose you perform a titration of an unknown weak acid solution. You start with 4.00 mL of the weak...
Questions

Computers and Technology, 22.03.2020 02:15


Mathematics, 22.03.2020 02:15

Mathematics, 22.03.2020 02:15



Mathematics, 22.03.2020 02:16

Mathematics, 22.03.2020 02:16

History, 22.03.2020 02:16


Mathematics, 22.03.2020 02:16

Mathematics, 22.03.2020 02:16




Mathematics, 22.03.2020 02:17



Mathematics, 22.03.2020 02:17

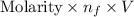
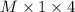 .......... (1)
.......... (1)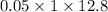 ............ (2)
............ (2)