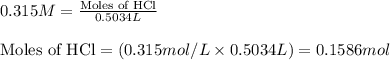Chemistry, 04.03.2020 01:28 masonrochester7
A mixture of hydrochloric and sulfuric acids is prepared so that it contains 0.315 M HCl and 0.125 M H2SO4. What volume of 0.55 M NaOH would be required to completely neutralize all of the acid in 503.4 mL of this solution?
hints
involves a solution that has 2 different acids in it. One way to do this is to imagine that you are neutralizing 2 solutions, one of each acid, and then just add the amounts of base needed. Another way is to think about how many moles of H3O+ are present in the mixed acids, and then figure out how much of the basic solution is needed to react with that amount of H3O+.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:30
What is the percentage by mass of silicon (si) in iron aluminum silicate (fe3al2(sio4)3)?
Answers: 2

Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1


Chemistry, 23.06.2019 03:00
Analyze the reaction to determine whether the reaction is exothermic or endothermic. explain your reasoning.
Answers: 1
You know the right answer?
A mixture of hydrochloric and sulfuric acids is prepared so that it contains 0.315 M HCl and 0.125 M...
Questions

Mathematics, 14.08.2020 02:01



History, 14.08.2020 02:01

Mathematics, 14.08.2020 02:01



Mathematics, 14.08.2020 02:01

Mathematics, 14.08.2020 02:01

Physics, 14.08.2020 02:01


Chemistry, 14.08.2020 02:01

Mathematics, 14.08.2020 02:01






Mathematics, 14.08.2020 02:01

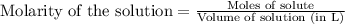 .....(1)
.....(1)