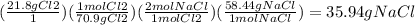Chemistry, 04.03.2020 01:16 ghari112345
In the complete reaction of 34.6 g of sodium with 21.8 g of chloride, what mass of sodium chloride is formed?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 2

Chemistry, 23.06.2019 02:00
Scientists are often interested in knowing the molar heat of combustion – the heat released during the combustion of one mole of a substance. use the periodic table to find molar masses. how many moles of ethanol are present in the sample?
Answers: 2
You know the right answer?
In the complete reaction of 34.6 g of sodium with 21.8 g of chloride, what mass of sodium chloride i...
Questions

Mathematics, 11.07.2019 08:50


History, 11.07.2019 08:50


Health, 11.07.2019 08:50



Chemistry, 11.07.2019 08:50

History, 11.07.2019 08:50




Mathematics, 11.07.2019 08:50

Advanced Placement (AP), 11.07.2019 08:50


English, 11.07.2019 08:50

Mathematics, 11.07.2019 08:50



Chemistry, 11.07.2019 08:50