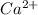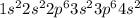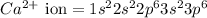Chemistry, 03.03.2020 19:51 Michael845313
The following Lewis diagram represents the valence electron configuration of a main-group element. This element is in group 2A According to the octet rule, this element would be expected to form a(n) with a charge of cation anion If X is in period 4, the ion formed has the same electron configuration as the noble gas The symbol for the ion is

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:30
**40** points asapessay questions (10 points possible) clear image of next, create your own scenario. it can be one of your own real experiences or one you make up. use imagery in your writing to give your instructor a the setting and an action taking pace in your writing explain the structure and functions of the skin at work in your scenario. !
Answers: 3

Chemistry, 21.06.2019 18:30
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible?
Answers: 2

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1
You know the right answer?
The following Lewis diagram represents the valence electron configuration of a main-group element. T...
Questions



Mathematics, 29.01.2020 15:01


Biology, 29.01.2020 15:01

Biology, 29.01.2020 15:01



Mathematics, 29.01.2020 15:01



Health, 29.01.2020 15:01


Mathematics, 29.01.2020 15:01


Chemistry, 29.01.2020 15:01