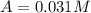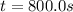Chemistry, 03.03.2020 06:07 Flowershere121
An average reaction rate is calculated as the change in the concentration of reactants or products over a period of time in the course of the reaction. An instantaneous reaction rate is the rate at a particular moment in the reaction and is usually determined graphically.
The reaction of compound A forming compound B was studied and the following data were collected:
Time (s) [A](M)
0. 0.184
200. 0.129
500. 0.069
800. 0.031
1200. 0.019
1500. 0.016
a.) What is the average reaction rate between 0. and 1500. s?
b.) What is the average reaction rate between 200. s and 1200. s?
c.) What is the instantaneous rate of the reaction at t=800 s?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 15:00
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1

Chemistry, 22.06.2019 23:00
What is the most common reason for matter changing its state?
Answers: 1

Chemistry, 23.06.2019 05:40
The independent variable in an experiment will be the variable that you o a) change ob) hold constant ng c) observe for changes
Answers: 2
You know the right answer?
An average reaction rate is calculated as the change in the concentration of reactants or products o...
Questions

History, 20.09.2019 03:40

Chemistry, 20.09.2019 03:40



Mathematics, 20.09.2019 03:40

Geography, 20.09.2019 03:40

Mathematics, 20.09.2019 03:40

Health, 20.09.2019 03:40

Biology, 20.09.2019 03:40


Mathematics, 20.09.2019 03:40

Mathematics, 20.09.2019 03:40


Spanish, 20.09.2019 03:40

History, 20.09.2019 03:40

Biology, 20.09.2019 03:40



Spanish, 20.09.2019 03:40

Mathematics, 20.09.2019 03:40

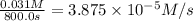
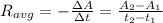
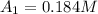
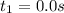
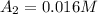
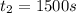
![R_{avg]=-\frac{0.016 M-0.184 M}{1500.0 s-0.0 s}=0.000112 M/s](/tpl/images/0531/9907/d3e1c.png)
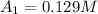
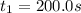
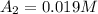
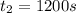
![R_{avg]=-\frac{0.019 M-0.129M}{1200.0s-200.0s}=0.00011 M/s](/tpl/images/0531/9907/ebcb3.png)