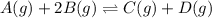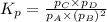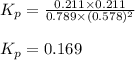Answers: 2
Another question on Chemistry



Chemistry, 22.06.2019 02:30
Based on the equation and the information in the table, what is the enthalpy of the reaction?
Answers: 2

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2
You know the right answer?
A(g) + 2B(g) → C(g) + D(g)If you initially start with 1.00 atm of both A and B and find that at equi...
Questions


Spanish, 28.08.2019 05:30


Mathematics, 28.08.2019 05:30


Health, 28.08.2019 05:30



Mathematics, 28.08.2019 05:30


Physics, 28.08.2019 05:30


Mathematics, 28.08.2019 05:30

Business, 28.08.2019 05:30

Biology, 28.08.2019 05:30

Mathematics, 28.08.2019 05:30



Social Studies, 28.08.2019 05:30

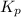 for the reaction is 0.169
for the reaction is 0.169