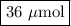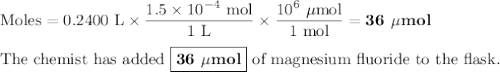Chemistry, 03.03.2020 03:26 ItzAquaZ8716
A chemist adds 240.0mL of a 1.5 x 10^-4 mol/L magnesium flouride (MgF2) solution to a reaction flask. Calculate the micromoles of magnesium fluoride the chemist has added to the flask. Be sure your answer has the correct number of significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:30
What is the force of attraction between the particles in a salt crystal
Answers: 2

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1

Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2
You know the right answer?
A chemist adds 240.0mL of a 1.5 x 10^-4 mol/L magnesium flouride (MgF2) solution to a reaction flask...
Questions

Mathematics, 14.04.2020 02:22


Mathematics, 14.04.2020 02:22

Mathematics, 14.04.2020 02:22



Mathematics, 14.04.2020 02:22





Mathematics, 14.04.2020 02:22

Mathematics, 14.04.2020 02:22

Physics, 14.04.2020 02:22

English, 14.04.2020 02:22

Mathematics, 14.04.2020 02:22


History, 14.04.2020 02:22


Mathematics, 14.04.2020 02:23