
Chemistry, 03.03.2020 02:52 wedestttefera
Consider the following chemical reaction: 2H2O(l)→2H2(g)+O2(g) What mass of H2O is required to form 1.3 L of O2 at a temperature of 295 K and a pressure of 0.926 atm ?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3
You know the right answer?
Consider the following chemical reaction: 2H2O(l)→2H2(g)+O2(g) What mass of H2O is required to form...
Questions

Chemistry, 20.10.2019 05:20

Mathematics, 20.10.2019 05:20


Mathematics, 20.10.2019 05:20


Computers and Technology, 20.10.2019 05:20

Mathematics, 20.10.2019 05:20

Physics, 20.10.2019 05:20

History, 20.10.2019 05:20

English, 20.10.2019 05:20

Geography, 20.10.2019 05:20





History, 20.10.2019 05:20

Mathematics, 20.10.2019 05:20


Chemistry, 20.10.2019 05:20

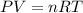
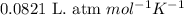

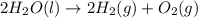
 of water
of water



