
Chemistry, 02.03.2020 22:42 shelbycg02
When 1.00 g of thiamine hydrochloride (also called vitamin B1 hydrochloride) is dissolved in water, then diluted to 10.00 mL, the pH of the resulting solution is 4.50. The formula weight of thiamine hydrochloride is 337.3 g/mol. Calculate Ka.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 10:00
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1

Chemistry, 22.06.2019 16:50
Ajet plane is speeding down the runway during takeoff. air resistance is not negligible. identify the forces on the jet.
Answers: 3
You know the right answer?
When 1.00 g of thiamine hydrochloride (also called vitamin B1 hydrochloride) is dissolved in water,...
Questions

Mathematics, 18.10.2021 05:10

History, 18.10.2021 05:10


History, 18.10.2021 05:10



Health, 18.10.2021 05:10


Mathematics, 18.10.2021 05:10


Mathematics, 18.10.2021 05:10

English, 18.10.2021 05:10

Mathematics, 18.10.2021 05:10

Mathematics, 18.10.2021 05:10


Mathematics, 18.10.2021 05:10


Geography, 18.10.2021 05:20

English, 18.10.2021 05:20

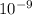
![\frac{[H+] [A-]}{[HA]}](/tpl/images/0530/9044/5a54d.png)
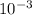
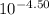
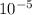
![\frac{[0.0000316] [0.000316]}{0.296}](/tpl/images/0530/9044/484a7.png)


