
Chemistry, 02.03.2020 21:23 kaykayhodge
He balanced net ionic equation for precipitation of CaCO 3 when aqueous solutions of Na 2CO 3 and CaCl 2 are mixed is . 2Na (aq) CO32- (aq) Na2CO3 (aq) 2Na (aq) 2Cl- (aq) 2NaCl (aq) Na (aq) Cl- (aq) NaCl (aq) Ca (aq) CO32- (aq) CaCO3 (s) Na2CO3 (aq) CaCl2 (aq) 2NaCl (aq) CaCO3 (s)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Bohr's model could only explain the spectra of which type of atoms? single atoms with one electron single atoms with more than one electron bonded atoms with one electron bonded atoms with more than one electron
Answers: 2

Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 05:00
When you mate two plants together the terms is called? answer it fast as possible plz! i have a test tomorrow!
Answers: 1

Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3
You know the right answer?
He balanced net ionic equation for precipitation of CaCO 3 when aqueous solutions of Na 2CO 3 and Ca...
Questions

History, 09.11.2020 19:40


Mathematics, 09.11.2020 19:40

Social Studies, 09.11.2020 19:40

Arts, 09.11.2020 19:40




English, 09.11.2020 19:40

Arts, 09.11.2020 19:40

Mathematics, 09.11.2020 19:40

Mathematics, 09.11.2020 19:40



Mathematics, 09.11.2020 19:40




Mathematics, 09.11.2020 19:40

Mathematics, 09.11.2020 19:40

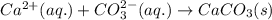
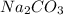 and
and 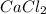 is an example of double decomposition reaction where each cations and anions of each salt exchange their partners during reaction to form
is an example of double decomposition reaction where each cations and anions of each salt exchange their partners during reaction to form 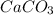 and NaCl
and NaCl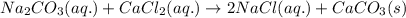
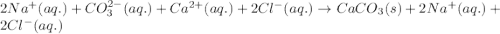
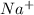 and
and 

