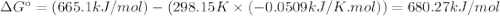Chemistry, 02.03.2020 21:22 jocelynmarquillo1
"Thermite" reactions have been used for welding metal parts such as railway rails and in metal refining. One such thermite reaction is 3 Mg(s) + Cr2O3(s) → 3 MgO(s) + 2 Cr(s). During the reaction, the surroundings absorb 665.1 kJ/mol of heat. Is the reaction spontaneous at room temperature (298.15 K) under standard conditions?

Answers: 2
Another question on Chemistry


Chemistry, 21.06.2019 19:00
Agood hypothesis includes which of the following? a: prediction b: data c: uncertainty d: conclusion
Answers: 1

Chemistry, 22.06.2019 07:30
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2

Chemistry, 22.06.2019 15:00
How is the shape of the poem “peer” connected to its meaning?
Answers: 2
You know the right answer?
"Thermite" reactions have been used for welding metal parts such as railway rails and in metal refin...
Questions








Chemistry, 28.10.2019 02:31

Health, 28.10.2019 02:31

History, 28.10.2019 02:31


History, 28.10.2019 02:31


Mathematics, 28.10.2019 02:31


History, 28.10.2019 02:31

Mathematics, 28.10.2019 02:31


Geography, 28.10.2019 02:31

Physics, 28.10.2019 02:31

![\Delta S^o_{rxn}=\sum [n\times \Delta S^o_{(product)}]-\sum [n\times \Delta S^o_{(reactant)}]](/tpl/images/0530/7449/52737.png)
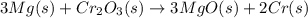
![\Delta S^o_{rxn}=[(3\times \Delta S^o_{(MgO(s))})+(2\times \Delta S^o_{(Cr(s))})]-[(3\times \Delta S^o_{(Mg(s))})+(1\times \Delta S^o_{(Cr_2O_3(s))})]](/tpl/images/0530/7449/80d1e.png)
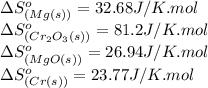
![\Delta S^o_{rxn}=[(3\times (26.94))+(2\times (23.77))]-[(3\times (32.68))+(1\times (81.2))]\\\\\Delta S^o_{rxn}=-50.88J/K=-0.0509kJ/K.mol](/tpl/images/0530/7449/fa231.png)
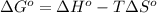
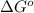 = standard Gibbs free energy = ?
= standard Gibbs free energy = ?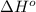 = standard enthalpy change of the reaction = 665.1 kJ/mol
= standard enthalpy change of the reaction = 665.1 kJ/mol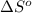 = standard entropy change of the reaction = -0.0509 kJ/K.mol
= standard entropy change of the reaction = -0.0509 kJ/K.mol