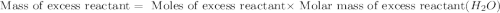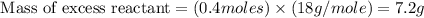Chemistry, 02.03.2020 18:20 dakshshberry
Onsider the following reaction: CaCN2 + 3 H2O → CaCO3 + 2 NH3 105.0 g CaCN2 and 78.0 g H2O are reacted. Assuming 100% efficiency, which reactant is in excess and how much is leftover? The molar mass of CaCN2 is 80.11 g/mol. The molar mass of CaCO3 is 100.09 g/mol.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Main expenses you plan on making payments on a new car too. you want to spend 15% of your monthly net pay on the car payment, insurance, registration, and taxes combined. what is your monthly car allowance? $149.46 $298.91 $448.37 $597.83
Answers: 3

Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2


Chemistry, 23.06.2019 01:30
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1
You know the right answer?
Onsider the following reaction: CaCN2 + 3 H2O → CaCO3 + 2 NH3 105.0 g CaCN2 and 78.0 g H2O are react...
Questions











Geography, 11.10.2019 00:00


Social Studies, 11.10.2019 00:00



History, 11.10.2019 00:00

Chemistry, 11.10.2019 00:00

Business, 11.10.2019 00:00


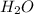
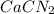 = 105.0 g
= 105.0 g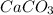 = 100.09 g/mole
= 100.09 g/mole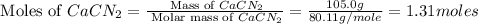
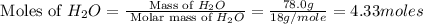
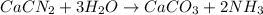
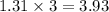 moles of
moles of