
Chemistry, 02.03.2020 18:18 cwsmith8026
. When iron rusts in air, iron(III) oxide is produced. How many moles of oxygen react with 2.4 mol of iron in the rusting reaction? 4Fe( s ) + 3O 2 ( g ) → 2Fe 2 O 3 ( s )

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1


Chemistry, 23.06.2019 01:30
Use the periodic table to determine how many grams of oxygen would be required to react completely with 859.0 g c2h2
Answers: 3

You know the right answer?
. When iron rusts in air, iron(III) oxide is produced. How many moles of oxygen react with 2.4 mol o...
Questions



Mathematics, 03.03.2021 19:00

Mathematics, 03.03.2021 19:00

History, 03.03.2021 19:00



Physics, 03.03.2021 19:00

Mathematics, 03.03.2021 19:00

Mathematics, 03.03.2021 19:00

Mathematics, 03.03.2021 19:00




Chemistry, 03.03.2021 19:00

Biology, 03.03.2021 19:00

Arts, 03.03.2021 19:00

Mathematics, 03.03.2021 19:00


Chemistry, 03.03.2021 19:00

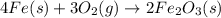
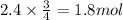 of oxygen gas.
of oxygen gas.

