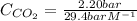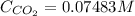Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:50
What are the 4 phases of matter in order of increasing engery content?
Answers: 2

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 16:30
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1

Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1
You know the right answer?
The Henry's law constant for CO 2 ( g ) CO2(g) in water at 25 ∘ C 25 ∘C is 29.4 bar⋅M − 1 29.4 bar·M...
Questions




Computers and Technology, 25.07.2019 05:00

Mathematics, 25.07.2019 05:00




English, 25.07.2019 05:00


English, 25.07.2019 05:00

Physics, 25.07.2019 05:00



Mathematics, 25.07.2019 05:00

Social Studies, 25.07.2019 05:00


Mathematics, 25.07.2019 05:00


Mathematics, 25.07.2019 05:00

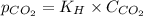
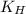 = Henry's constant =
= Henry's constant = 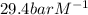
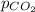 = partial pressure of carbonated drink = 2.20 bar
= partial pressure of carbonated drink = 2.20 bar